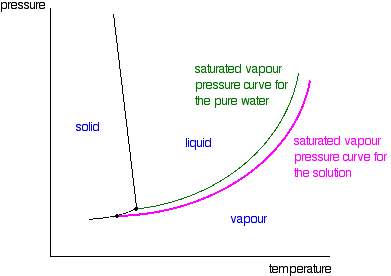
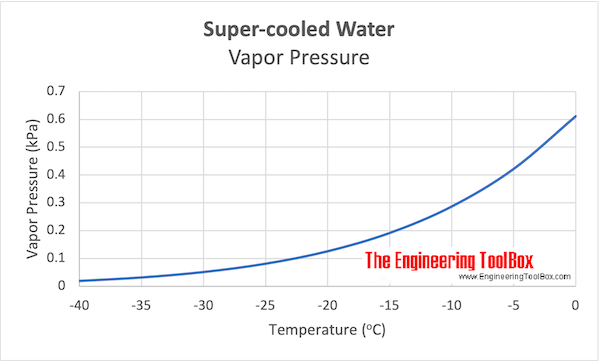
The vapour pressure of pure water at 20^oC is 20 mm Hg . A solution of sucrose whose molar mass is 342 g/mole is prepared by dissolving 75 g of it in

SOLVED: Question 10 (1 point) The vapor pressure of pure water at 50 %C is 92.5 mmHg: A solution containing the non-electrolyte sucrose (MM = 342 g/mol) has vapor pressure of 90.8

vapor pressure - Why is water transferred from a beaker of pure water to a beaker of sugar solution when placed in the a closed container? - Chemistry Stack Exchange

Saturation pressure of pure water and concentrated brine as a function... | Download Scientific Diagram

SOLVED: The vapor pressure of pure water at 25°C is 24.0 mm Hg. What is the expected vapor pressure at 25°C of an ideal solution of a nonvolatile nonelectrolyte in which the

Find the molality of a solution containing a non - volatile solute if the vapour pressure is 2% below the vapour pressure of pure water.

When considering the vapor pressure of pure water and seawater at constant temperature, which of the following statements are correct? a. Seawater will have a higher vapor pressure than water. b. Water

The vapour pressure of pure water at `25^(@)"C"` is 23.76 torr. The vapour pressure of a solution - YouTube

Vapour pressure of pure water at `298 K ` is `23.8 mm Hg`. `50g` of urea `(NH_(2)CONH_(2))` is - YouTube




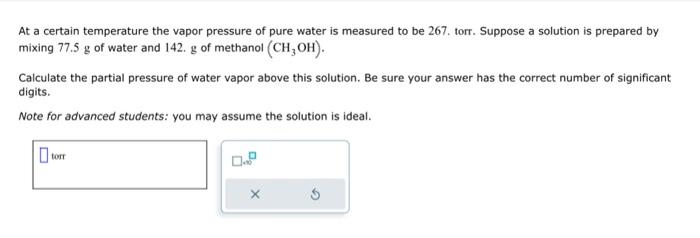


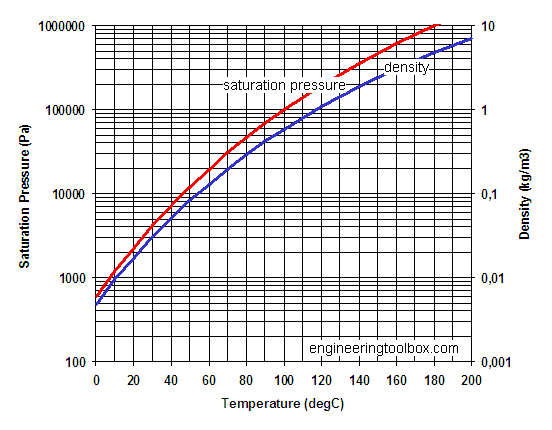
![PDF] Saturated vapor pressures over Great Salt Lake brine | Semantic Scholar PDF] Saturated vapor pressures over Great Salt Lake brine | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/fe823e993367e8a74e59d6788ad2394e689a48cf/2-Table2-1.png)