
Novavax COVID-19 vaccine researched in Oxford approved in UK by MHRA - Oxford Health NHS Foundation Trust
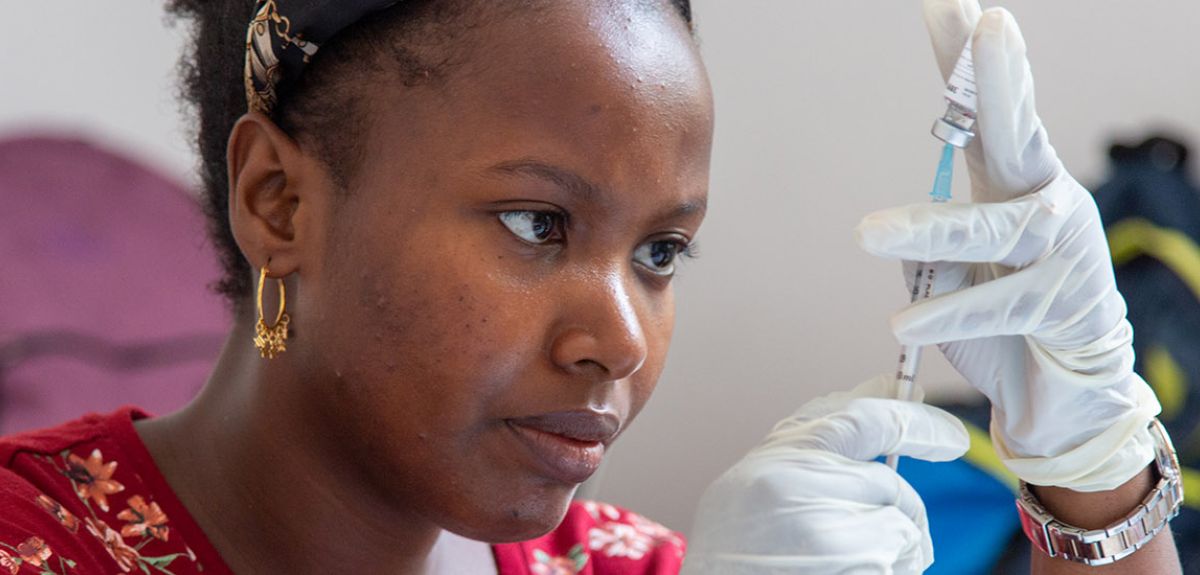
Oxford R21/Matrix-M™ malaria vaccine receives WHO recommendation for use paving the way for global roll-out | University of Oxford

Novavax's long-awaited COVID-19 vaccine authorizations offer an alternative to mRNA | Science | AAAS

World Health Organization (WHO) on X: "WHO's emergency use listing procedure assesses the quality, safety and efficacy of #COVID19 vaccines and is a prerequisite for #COVAX vaccine supply. It also allows countries

WHO lists 9th COVID-19 vaccine for emergency use with aim to increase access to vaccination in lower-income countries

Novavax on X: "NEWS RELEASE: Novavax and @SerumInstIndia Announce Submission to the World Health Organization for Emergency Use Listing of Novavax' COVID-19 Vaccine Candidate https://t.co/MFTIxPgroV https://t.co/OHgpv2o2kz" / X
WHO lists 10th COVID-19 vaccine for emergency use : Nuvaxovid | Strategic Partnership for Health Security and Emergency Preparedness (SPH) Portal











:max_bytes(150000):strip_icc()/Novavax-Files-for-FDA-to-Authorize-Their-COVID-19-Vaccine-Heres-What-to-Know-AdobeStock_476294316_Editorial_Use_Only-2000-3dbc509f2c2a4d5985d8e372cc736433.jpg)