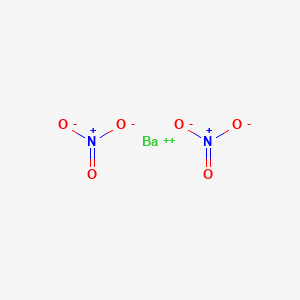
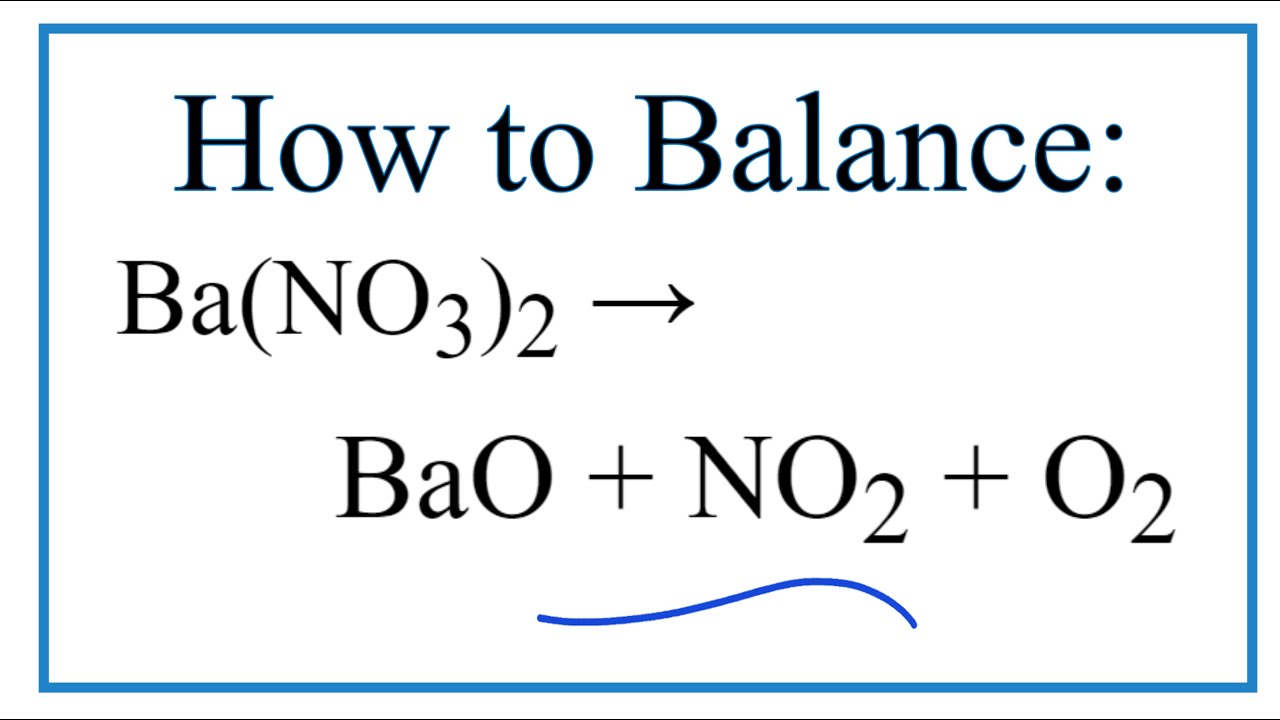
Barium Nitrate : United Nuclear , Scientific Equipment & Supplies, United Nuclear , Scientific Equipment & Supplies

White Barium Nitrate For Multipurpose Use Application: Industrial at Best Price in Ahmedabad | Usha Enterprise

Barium Nitrate, 99.9999% (Metals Basis), Certified, 25g (Hazmat Fee Included in Price) - Chemsavers, Inc.

Barium Nitrate and Nitric Acid Barium Chloride Plus Sodium Carbonate Natural Barium Sulphate Barium Sulfate Pigment - China Barium Nitrate and Nitric Acid, Barium Chloride Plus Sodium Carbonate | Made-in-China.com

Barium Nitrate Molecular Structure Formula Periodic Stock Vector (Royalty Free) 2305779911 | Shutterstock



















![Barium nitrate - Optional[Raman] - Spectrum - SpectraBase Barium nitrate - Optional[Raman] - Spectrum - SpectraBase](https://spectrabase.com/api/spectrum/3qvrObeRn13/partial.png?h=214.875&ph=true&w=382)