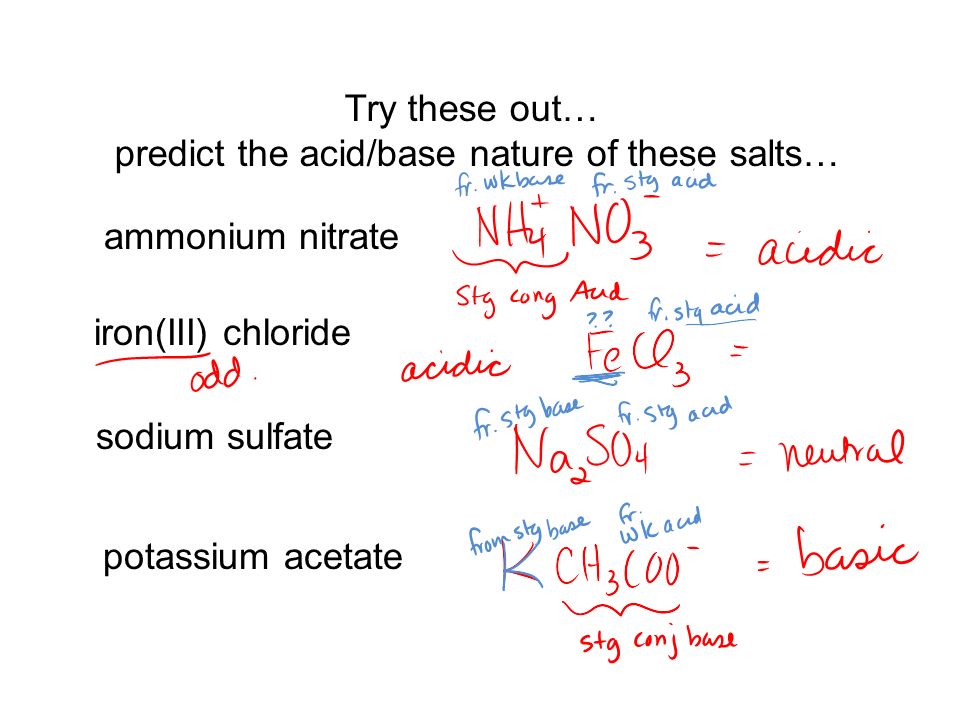
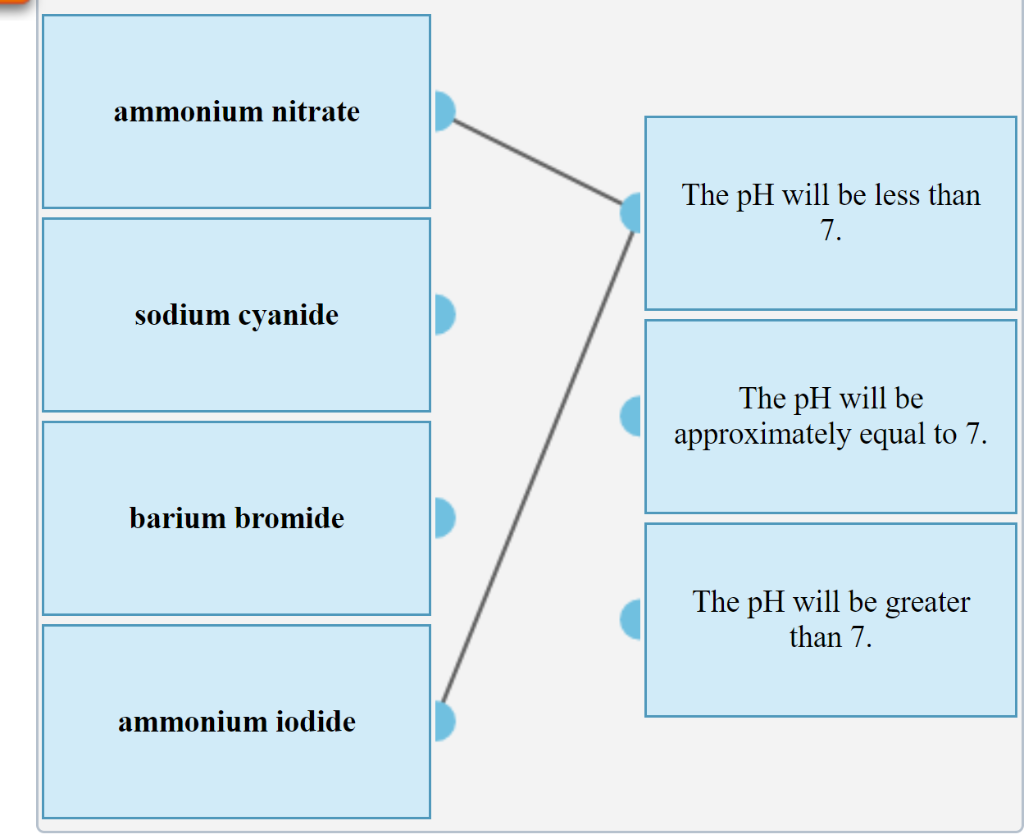
SOLVED: Nitric acid (HNO 3 ) reacts with ammonia (NH 3 ) to yield ammonium nitrate. Write the reaction, and identify the acid, the base, the conjugate acid product, and the conjugate base product.
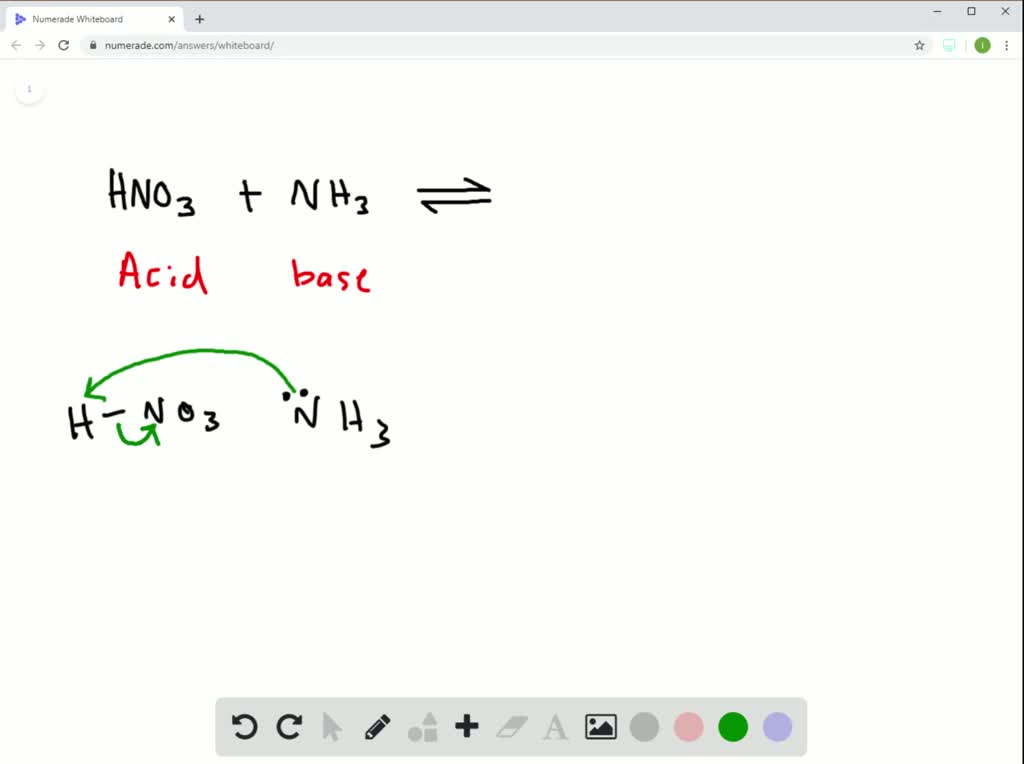
SOLVED:Nitric acid (HNO 3 ) reacts with ammonia (NH 3 ) to yield ammonium nitrate. Write the reaction, and identify the acid, the base, the conjugate acid product, and the conjugate base product.

Name the acid and base needed to form the salt ammonium nitrate give the chemical reaction - Brainly.in

Ammonium nitrate, NH4NO3, is a salt formed from the neutralization of the weak base ammonia with the strong acid nitric acid. Given that the value of Kb for ammonia is 1.8 x

Ammonium Nitrate (NH4NO3) - Formula, Structure, Preparation, Physical and Chemical Properties, Uses with FAQs of Ammonium Nitrate

Displacement of Strong Acids or Bases by Weak Acids or Bases in Aerosols: Thermodynamics and Kinetics | Environmental Science & Technology