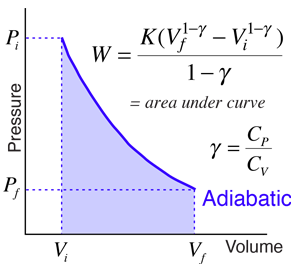
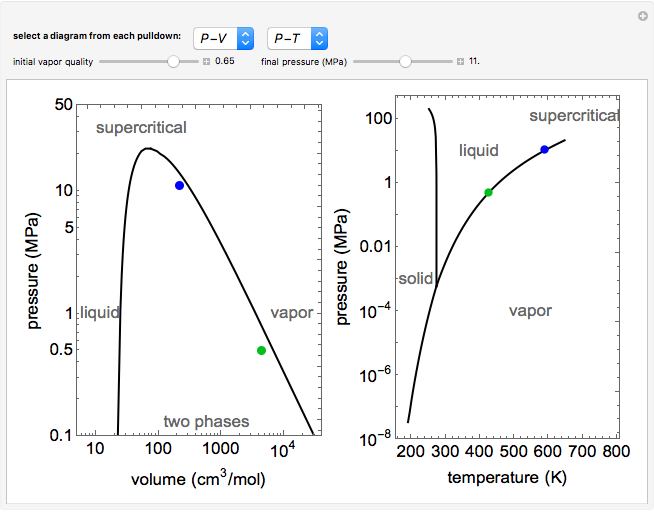
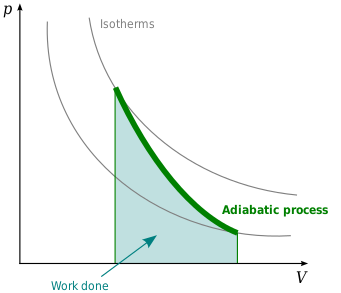
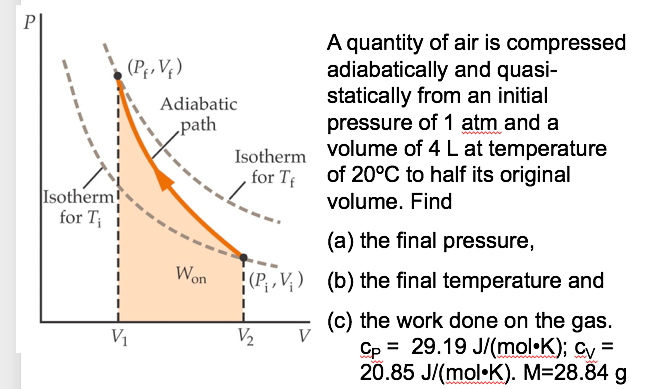
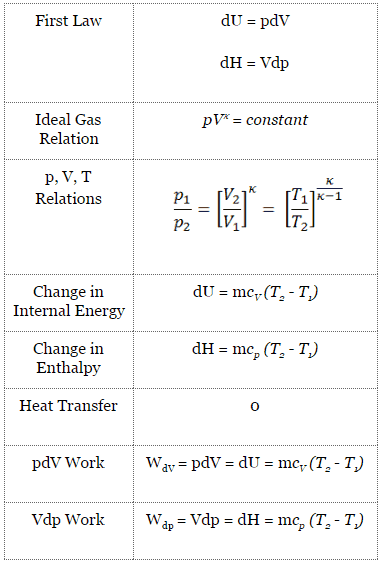Argon gas is adiabatically compressed to half its volume. If P, V and T represent the pressure, ... - YouTube
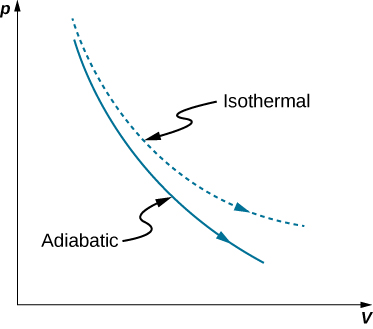
2.5 Adiabatic Processes: The Path of Least Resistance | METEO 300: Fundamentals of Atmospheric Science
An ideal monatomic gas is adiabatically compressed so that its final temperature is twice its initial temperature. - Sarthaks eConnect | Largest Online Education Community

Air is first compressed adiabatically from 100 kPa and 300 K to 1500 kPa and a specific volume of 0.1 m3/kg. The air is then cooled at constant volume back to 300

an ideal gas is compressed to half its initial volume The same gas is compressed separately through an adiabatic - Physics - Kinetic Theory - 11986627 | Meritnation.com


![Bengali] Some amount of air, initially at STP, is adiabatically compr Bengali] Some amount of air, initially at STP, is adiabatically compr](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/3845343.webp)